The QM solution for your pharmacy
The QM solution for your pharmacy









By digitizing our QM documentation with roXtra, we can optimally prepare audits for our pharmacies.
Philipp Wälde, Owner BLESS YOU Pharmacies
Success Stories
Find out first-hand how users use roXtra to design their quality management systems (QMS) efficiently and in compliance with regulations. Whether ISO 9001, ISO 13485 or the QM guidelines of the German Federal Chamber of Pharmacists (BAK) - roXtra offers solutions that are specially tailored to the requirements of pharmacies.
In our blog, you can find out more about certification and re-certification, the implementation of quality management in pharmacies and practical application examples from the industry.

Special requirements for QM software
for decentralized project processes
Asklepios clinics
Get advice now
Get to know roXtra in a non-binding and free online presentation.
What is a quality management system in a pharmacy?
Quality management in pharmacies (QM) encompasses all measures that ensure consistently high quality in the manufacture, testing and dispensing of medicines. This includes structured processes, complete documentation and regular audits in order to meet legal requirements and customer demands. Powerful QM software supports pharmacies in document control, process management, audit planning and risk management and facilitates the implementation of continuous improvement measures.
Modern software solutions for quality management in pharmacies automate processes and ensure greater transparency and efficiency. This minimizes sources of error, optimizes work processes and makes it easier to comply with legal requirements.
What activities does quality management in pharmacies involve?
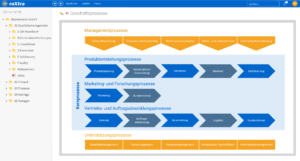
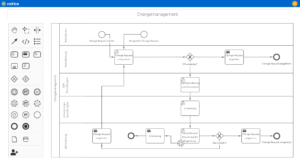
Quality management in pharmacies involves numerous tasks that serve both to comply with legal requirements and to optimize internal processes. This includes document control in order to keep all relevant work instructions, process descriptions and protocols up to date and audit-proof. Process management also plays a central role by defining standardized processes and ensuring compliance with them. Efficient QM software for pharmacies helps to manage these tasks in a structured and digital way.
Other important activities in quality management for pharmacies are audit management, which is used to plan and document internal and external audits. Risk management enables potential risks to be identified and minimized at an early stage. Systematic action management allows necessary improvements to be implemented quickly. Digital QM software solutions ensure efficient implementation of all QM processes and help pharmacies to permanently meet the high quality and safety requirements.
Why is quality management important in pharmacies?
Quality management in pharmacies is essential to ensure a safe and reliable supply of medicines. Standardized processes minimize errors, increase patient safety and comply with legal requirements. In addition, structured quality management helps to increase efficiency, improves customer satisfaction and promotes the continuous development of pharmacy processes. In an increasingly regulated and competitive environment, it ensures the long-term success and trustworthiness of the pharmacy.
How can a pharmacy manage its QM documents efficiently?
A pharmacy can manage its QM documents efficiently by using a digital document management solution that enables central storage, versioning and access control. Clear responsibilities and regular reviews ensure that all documents are up-to-date and legally compliant. In addition, a structured folder hierarchy or specialized QM software makes it easier to find and process relevant documents quickly. Training for employees and the integration of automated workflows also help to minimize administrative work and ensure compliance with quality standards.
How can a pharmacy carry out internal audits efficiently?
A pharmacy can carry out internal audits efficiently by using specialized audit software. This enables structured planning, implementation and documentation of audits. Digital checklists and automated reminders ensure a complete review of relevant processes, while integrated reporting and analysis functions quickly identify weaknesses. The software also makes it easier to track corrective measures and ensures transparent communication within the team. The central storage of all audit data reduces administrative effort and supports compliance with legal requirements in the long term.
One software, many application areas.
Risk management for pharmacies with roXtra Risks
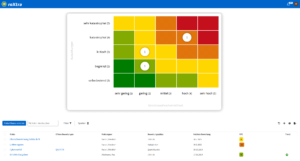
With roXtra Risks you minimize the effort involved in risk management. Identify, analyze and manage potential risks centrally to ensure safety and quality in your pharmacy. The QM software supports regulatory requirements such as the German Pharmacy Operating Regulations (ApBetrO), GMP and ISO 9001 and facilitates the implementation of effective quality management in pharmacies.
Effective risk management for maximum safety
Regulatory requirements demand structured risk management in order to identify risks such as incorrect dispensing of medicines or hygiene deficiencies at an early stage. The QM software from roXtra helps you to systematically assess risks, initiate measures and continuously optimize your QM processes.
roXtra supports you with the following solutions
We will show you roXtra in a free and non-binding online presentation.






