Your solution for quality management in medicine
Your solution for quality management in medicine
Already in use in over two thirds of German university hospitals
: our software solutions









Success Stories
Find out first-hand how university hospitals use roXtra to make quality management in medicine efficient and compliant. Whether ISO 9001, KTQ or comprehensive risk management - roXtra offers customized solutions for structured and standard-compliant medical quality management that are specially tailored to the requirements of hospital operations.
In our blog, you can find out more about certification and re-certification, the implementation of quality management in medicine and practical application examples from the industry.
Get advice now
Get to know roXtra in a non-binding and free online presentation.
Why does medicine need quality management?
Quality management in medicine is essential to ensure safe, efficient and patient-oriented care. Structured medical quality management helps university hospitals to comply with legal requirements and medical standards, optimize processes and minimize risks.
In addition, efficient quality management helps to increase patient safety, drive continuous improvements and overcome economic and organizational challenges in modern medicine.
What does quality management do for medicine?
Effective and structured quality management in medicine ensures transparent processes in university hospitals. It includes compliance with legal requirements, the optimization of clinical processes and the continuous improvement of various standards. Effective medical quality management minimizes risks, increases patient safety and supports specialists in carrying out their work efficiently and in compliance with regulations.
What quality management regulations must university hospitals comply with?
University hospitals in Germany are subject to various quality management regulations. Central to this are the provisions of the German Social Code (SGB V, § 135a and § 137), which prescribe continuous quality assurance and reporting. The Federal Joint Committee (G-BA) also stipulates specific guidelines, including the guideline on cross-institutional quality assurance (Qesü-RL). Many clinics follow the international standard DIN EN ISO 9001 or are certified according to the KTQ procedure (Cooperation for Transparency and Quality in Healthcare).
In addition, university hospitals must comply with EU regulations such as the Medical Device Regulation (MDR), particularly when dealing with medical devices and clinical research. In addition, occupational health and safety and hygiene regulations, including the Infection Protection Act (IfSG) and the recommendations of the Robert Koch Institute (RKI), govern key aspects of patient safety. Many hospitals are also developing internal quality management systems to ensure the highest standards of patient care.
How does QM in a university hospital differ from that in a normal hospital?
Quality management (QM) in a university hospital differs from that in a normal hospital primarily due to the additional requirements for research and teaching. In addition to patient safety and medical care, which play a central role in both types of hospital, university hospitals must also meet high quality standards in clinical research and the training of medical students and specialist staff. These include compliance with Good Clinical Practice (GCP) in clinical trials, the Medical Device Regulation (MDR) for the safe use of medical devices and strict ethical and scientific standards.
In addition, university hospitals often work together with several external partners, such as research institutes and universities, which requires a particularly complex QM system. Furthermore, they are often certified according to specialized standards such as DIN EN 15224 or ISO 9001 and must meet the requirements of funding bodies and scientific societies in addition to legal requirements. Overall, QM in university hospitals is therefore more comprehensive, more multidisciplinary and more focused on innovation and knowledge transfer.
To what extent does QM influence the further training and qualification of medical staff?
Quality management (QM) ensures the further training and qualification of medical staff in university hospitals through fixed standards, regular training and certifications such as ISO 9001 or KTQ. Digital training platforms, on which employees can flexibly access training content, as well as standard operating procedures (SOPs) and simulation training ensure practical training. In addition, professional associations and state medical associations require certified further training. In this way, QM contributes to continuous improvement and safe patient care.
One software, many application areas.
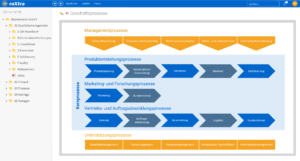
Effective document control
With roXtra document control, you can maintain an overview of your documentation in medical quality management at all times - from creation to archiving. Independent of location and always up-to-date, roXtra facilitates the structured checking and approval of documents. This ensures efficient medical quality management and transparency.
Your advantages with roXtra in the area of document control:
- Individual approval workflows: Customizable to your processes and standard requirements.
- Audit-proof versioning: automatic storage and archiving.
- Standard-compliant document management: For all directives, e.g. DIN EN 15224 or GxP.
- Rely on a future-proof software solution to make your quality management processes in medicine efficient, compliant and transparent.
Efficient and digital
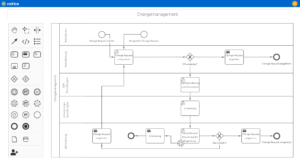
Digitize and automate your internal processes with the roXtra Processes module. Thanks to graphical modeling in accordance with BPMN 2.0, you can easily define and control rules, responsibilities, tasks and input fields for various processes and forms (e.g. patient information, treatment procedures).
roXtra Processes enables efficient, compliant and digital processes - perfectly adapted to the requirements of university hospitals.
With the Flowchart Designer, you can create simple models such as flowcharts, organization charts or mind maps in no time at all and make your processes easy to understand for everyone involved.
With the Flowchart Designer, you can create simple models such as flowcharts, organization charts or mind maps in no time at all and make your processes easy to understand for everyone involved.
Advantages of process management with roXtra:
- Transparency and clarity: Visualization of complex processes such as clinical trial procedures, certification processes or quality assurance measures.
- Continuous improvement (CIP): Optimize processes and make them sustainably efficient.
- User-friendliness: Intuitive design and fast implementation of your requirements.
Risk management for university hospitals
With roXtra Risks you can reduce your risk management workload and improve your risk analysis. Identify and manage potential risks centrally to ensure the safety and quality of your medical devices. The software supports common standards and regulations, including ISO 31000 and ONR 49001, ISO 15224, ISO 9001 and ISO 27001 / IEC 8001.
Your advantages with roXtra Risks:
- Efficient risk identification: identify and analyze risks at an early stage.
- Customization: Flexibly define risk categories and valuation cycles.
- Clear control: matrices and risk reports for comprehensive reporting.
- Rapid integration of measures: derive and link measures directly.
Effective audit management
Audits are an indispensable part of your work as a quality manager and internal auditor. Systematic audit management is your most powerful tool for checking processes, uncovering non-conformities and driving improvements.
With the module roXtra Audits module, you can increase the effectiveness and quality of your work. The software supports you in carrying out and managing internal audits and auditing management systems in accordance with specifications such as ISO 19011.
Whether DIN EN ISO 9001, ISO 13485, DIN EN ISO/IEC 17025 or ISO 14001 - with roXtra Audits you can easily meet the requirements of current standards. The software helps you to identify potential for improvement, systematically plan and carry out internal audits and ensure traceability.
Your advantages with roXtra Audits:
- Efficiency and transparency: Document and initiate immediate, corrective and preventive measures directly in the context of the audit.
- Systematic implementation: Access your questionnaire from any location - even offline.
- Optimized planning: Maintain an overview of past, current and planned audits.
Action management for university hospitals
Effective action management with roXtra Actions is crucial for eliminating deviations, minimizing risks and meeting the requirements of standards such as DIN EN ISO 9001 and DIN EN ISO 31000. With a structured approach, immediate, corrective and preventive actions (CAPA) can be efficiently planned, implemented and tracked.
A digitally supported system guarantees complete documentation and transparent monitoring of all measures, ensuring continuous improvement and compliance with regulatory requirements.
Your advantages with roXtra Actions:
- Central management: Clearly record and control all measures.
- Automatic effectiveness check: Sustainable implementation through planned checks.
- Simple evaluation: Filter lists of measures and export them as Excel, Word or PDF.
- Cross-module: Linking for consistent documentation.
Support for quality management and certifications in medical technology
Our software solutions make your daily operations as well as the certification and re-certification of your quality management system (QMS) easier. With roXtra, you can implement a wide range of standards and regulations efficiently and in compliance with the rules - from DIN EN 15224 to ISO 27001.
Quality management in medicine
- KTQ certification: Cooperation for transparency and quality in healthcare
- DIN EN 15224 Quality management systems for healthcare
- ISO 7101 Quality management systems in healthcare - Requirements
- DIN EN ISO 13485 Medical device quality management systems - Requirements for regulatory purposes
- Claims of the Joint Federal Committee (G-BA), the German Hospital Federation (DKG) and the German Nursing Council (DPR)
- NÖ BASIS ZERT
Risk management and security
- DIN EN ISO 31000 - Guidelines for risk management
- or ONR 49000 / ONR 49001 - Austrian risk management standard
- Requirements for operators of critical infrastructures (KRITIS)
- IEC 80001 standard for risk management in networked medical technology
- § Section 91 (2) AktG - Establishment of a risk management system (RMS) for listed companies
- KonTraG - Law on Control and Transparency in the Corporate Sector (Corporate Governance)
Quality management in general
- DIN EN ISO 9000 series of standards: Quality management standards
- DIN EN ISO 9001: Quality management systems for companies
- ISO 27001: Information security management system (ISMS) and IT security
roXtra supports you with the following solutions
We will show you roXtra in a free and non-binding online presentation.







